Exploring the Significance of Hydrogen Electrolysis
Exploring the Significance of Hydrogen Electrolysis https://h2-heat.eu/wp-content/uploads/2024/04/processes-08-01634-g001-1024x738.webp 1024 738 H2Heat Project https://h2-heat.eu/wp-content/uploads/2024/04/processes-08-01634-g001-1024x738.webpElectrolysis is a process that uses electricity to split substances into simpler parts. It’s like breaking down something big into smaller pieces.
Imagine you have a big water molecule (H2O). Electrolysis can split it into hydrogen (H2) and oxygen (O2). This splitting happens in a special device called an electrolytic cell. This cell has two electrodes and an electrolyte. The electrodes are like tiny metal sticks that conduct electricity. The electrolyte is a liquid or solution that conducts electricity too. When you pass electricity through the electrolyte, it causes a chemical reaction.
This reaction breaks down the substance into its components. At the positive electrode, called the anode, negatively charged ions or electrons are attracted. At the negative electrode, called the cathode, positively charged ions are attracted. The ions move towards the oppositely charged electrodes. At the electrodes, they react and form new substances. For example, in water electrolysis, at the anode, water molecules lose electrons and form oxygen gas and hydrogen ions.
At the cathode, hydrogen ions gain electrons and form hydrogen gas. Electrolysis is used in many industries, from producing metals like aluminium to purifying metals like copper. It’s also used to produce chemicals like chlorine and sodium hydroxide. In the future, electrolysis could play a big role in producing clean fuels like hydrogen, which can power vehicles and generate electricity without producing greenhouse gases.
The Significance of Hydrogen Electrolysis
Hydrogen electrolysis, also known as water electrolysis, is a groundbreaking process that involves the separation of water (H2O) into its constituent elements—hydrogen (H2) and oxygen (O2)—through the application of an electric current. This technology has gained significant attention as a key enabler of the transition towards a sustainable energy landscape.
Advantages of Hydrogen as an Energy Carrier
Hydrogen’s versatility lies in its ability to be produced using renewable energy sources such as wind, solar, and hydropower. This green hydrogen production holds immense potential for reducing the environmental footprint across diverse industries. By using renewable energy for electrolysis, hydrogen can serve as a clean and efficient energy carrier, facilitating the decarbonisation of various industrial processes.
Exploring Methods of Hydrogen Electrolysis
Overview of Hydrogen Electrolysis Methods
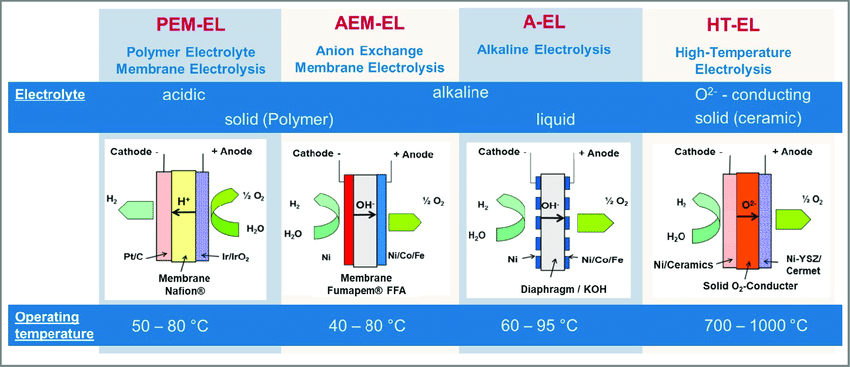
Hydrogen electrolysis encompasses three primary methods: alkaline electrolysis, proton exchange membrane (PEM) electrolysis, and solid oxide electrolysis. Each method offers unique advantages and applications, catering to diverse industrial requirements. While alkaline electrolysis is widely recognized for its scalability and cost-effectiveness, PEM electrolysis is prized for its efficiency and flexibility. Solid oxide electrolysis, on the other hand, boasts high-temperature operation and compatibility with heat sources such as nuclear energy.
Understanding Alkaline Electrolysis
Alkaline electrolysis stands as one of the most established methods of hydrogen production. This process entails the splitting of water molecules (H2O) into hydrogen (H2) and oxygen (O2) within an alkaline electrolytic cell. Employing an alkaline electrolyte solution, typically potassium hydroxide (KOH) or sodium hydroxide (NaOH), this method facilitates ion flow between electrodes to enable efficient hydrogen generation.
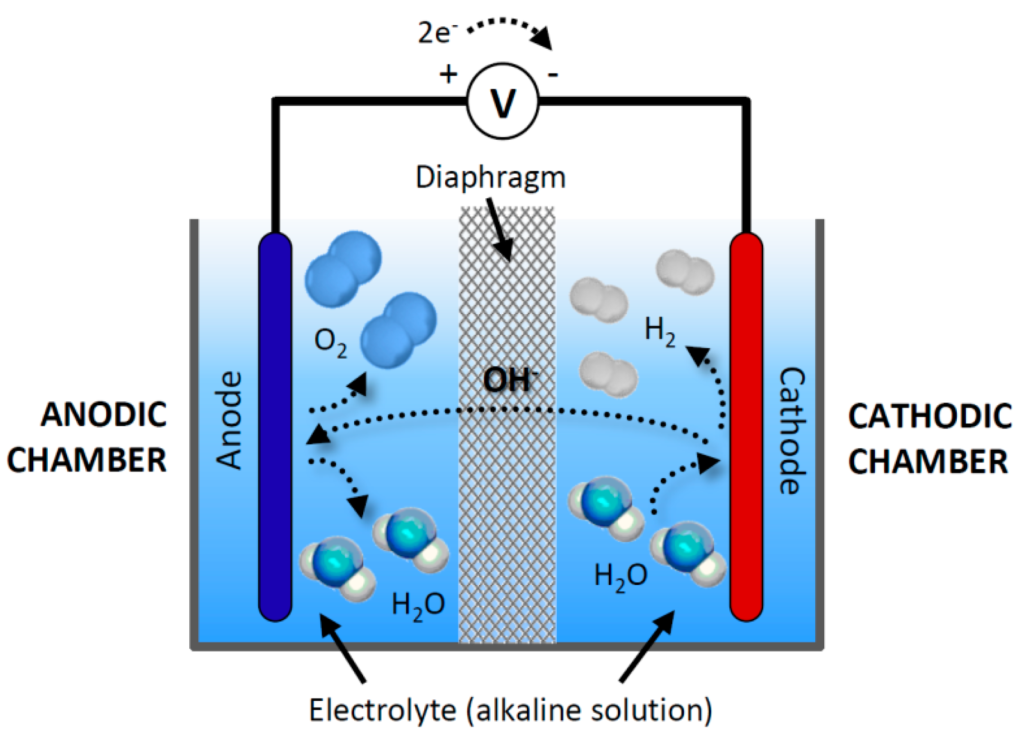
Components of an Alkaline Electrolysis Cell
The electrolysis cell comprises two electrodes—an anode and a cathode—immersed in the alkaline electrolyte solution. Typically crafted from durable and corrosion-resistant materials like nickel, these electrodes play a crucial role in the electrolysis process. As the electric current passes through the cell, water molecules undergo electrolysis, yielding hydrogen gas at the cathode and oxygen gas at the anode.
Hydrogen electrolysis is one of the clean energy technologies, offering a sustainable pathway towards decarbonising industrial processes and achieving environmental sustainability. As industries worldwide strive to embrace renewable energy solutions, hydrogen electrolysis emerges as a key enabler of this transition. By understanding the principles and applications of electrolysis, we can unlock the full potential of hydrogen as a clean and efficient energy carrier, driving positive change for generations to come.
Reshaping Industries for a Sustainable Future
The implications of hydrogen electrolysis extend far and wide, touching upon critical sectors that form the backbone of modern civilization.
Glass and Ceramics: In the glass and ceramics industries, hydrogen electrolysis presents an opportunity to reduce reliance on fossil fuels and transition towards cleaner production methods. By embracing hydrogen as an energy source, these sectors can mitigate their environmental footprint while maintaining operational efficiency.
Concrete Production: Concrete, a cornerstone of construction, traditionally relies on energy-intensive processes. However, with hydrogen electrolysis, concrete manufacturers can adopt greener practices, leading to lower emissions and a more sustainable construction sector.
Steelmaking: The steel industry, known for its significant carbon emissions, stands to benefit immensely from hydrogen electrolysis. By replacing traditional fossil fuels with hydrogen, steelmakers can drastically reduce their carbon footprint and embrace a more environmentally friendly approach to production.
Fertilisers: Agricultural fertilisers play a crucial role in global food production but often come with environmental drawbacks. Hydrogen electrolysis offers a pathway to producing ammonia – a key component of fertilizers – using clean energy sources. This shift could revolutionise the agriculture sector, paving the way for more sustainable farming practices.
